THRIVE
The Trajectories of Recover after Intravenous propotfol vs. inhaled VolatilE anesthesia trial is a multicenter, clinical trial evaluating the superiority of propofol vs.TIVA (inhaled volatile general anesthesia.
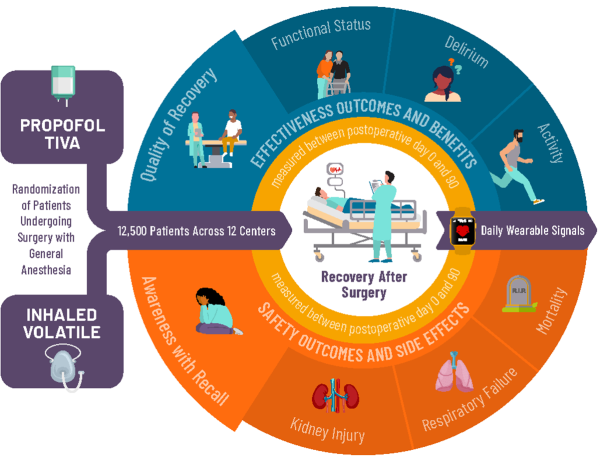
 THRIVE is a multicenter, patient-level, parallel group, pragmatic comparative effectiveness, randomized control trial evaluating the superiority of propofol TIVA over inhaled volatile general anesthesia. This initiative is supported by the Patient-Centered Outcomes Research Institute (PCORI). The project will leverage the existing MPOG infrastructure, with the University of Michigan serving as the Data Coordinating Center, and Washington University St. Louis serving as the Clinical Coordinating Center.
THRIVE is a multicenter, patient-level, parallel group, pragmatic comparative effectiveness, randomized control trial evaluating the superiority of propofol TIVA over inhaled volatile general anesthesia. This initiative is supported by the Patient-Centered Outcomes Research Institute (PCORI). The project will leverage the existing MPOG infrastructure, with the University of Michigan serving as the Data Coordinating Center, and Washington University St. Louis serving as the Clinical Coordinating Center.
The project is expected to last 6.5 years, during which time 12,500 patients will be enrolled at approximately 22 MPOG sites. Adult patients undergoing elective non-cardiac surgeries lasting longer than 60 minutes, using general anesthesia either as inpatients or ambulatory cases will be eligible.
By comparing propofol TIVA and inhaled volatile anesthesia, the THRIVE team hopes to improve patient outcomes, by determining whether one of the two tested general anesthesia techniques benefits quality of recovery and functional status, while assessing any impact on risk of delirium, intraoperative awareness with recall, kidney injury, respiratory failure and mortality.
Please review our other website pages to learn more about the structure of the THRIVE trial, view our timeline, read our monthly newsletters, and view additional clinician educational resources.
More information regarding becoming a THRIVE enrollment site can be found on the
Information for Interested Sites page.
For general THRIVE inquiries please contact askthrive@umich.edu. If you need to reach out to a specific THRIVE team member, please refer to
the Operational Structure webpage for contact information.

