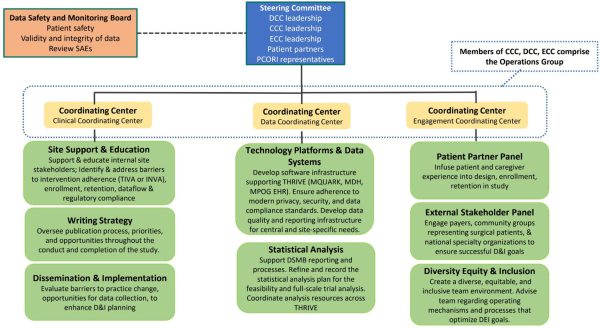
Administrative Structure
THRIVE is built on a multi‑layered, collaborative leadership and working‑group structure designed to drive coordination, accountability, and scientific rigor across every phase of the trial. This integrated framework brings together experts, stakeholders, and community partners to ensure that decision‑making is transparent, operations are efficient, and the trial is positioned for long‑term success.